FLUIMUCIL MUCOL * OS 10BUST600MG
Therapeutic indications
Treatment of respiratory diseases characterized by thick and viscous hypersecretion.
Dosage and method of use
Adults: 1 sachet of Fluimucil Mucolytic 200 mg granules for oral solution (with or without sugar) or 2 sachets of Fluimucil Mucolytic 100 mg (with or without sugar) 2-3 times a day. Fluimucil Mucolytic 200 mg, buccal tablets and effervescent tablets: 1 tablet 2-3 times a day. Fluimucil Mucolytic 100 mg / 5 ml, syrup: 10 ml of syrup (1 scoop), equal to 200 mg of N-acetylcysteine, 2-3 times a day. Fluimucil Mucolitico 600 mg / 15 ml syrup, Fluimucil Mucolitico 600 mg effervescent tablets and Fluimucil Mucolitico 600 mg granules for solution: a 15 ml measuring spoon or an effervescent tablet or a sachet (preferably in the evening). Possible dose adjustments may relate to the frequency of administration or dose fractionation but must still be within the maximum daily dosage of 600 mg.Children over 2 years of age: Fluimucil Mucolytic 100 mg granules for oral solution (with or without sugar): 1 sachet 2 to 4 times a day, according to age. Fluimucil Mucolytic 100 mg / 5 ml, syrup: ½ measuring spoon of syrup (5 ml), equal to 100 mg of N-acetylcysteine, 2 to 4 times a day according to age. The duration of therapy is from 5 to 10 days.Method of administration Granules for oral solution: dissolve the contents of one sachet in a glass containing a little water, stirring as needed with a teaspoon. In this way a pleasant solution is obtained which can be drunk directly from the glass or, in the case of small children, be given in teaspoons or in a bottle. The solution should be taken as soon as it is ready.Buccal tablets: keep the tablet in the oral cavity until it is completely dissolved.Syrup: shake before using. Once opened, the syrup is valid for 15 days. Effervescent tablets: dissolve one tablet in a glass containing a little water, stirring as needed with a teaspoon. To facilitate the release of the tablet, we recommend the tear-off opening of the blister, using the side notches as shown in the figure.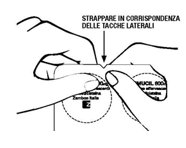
Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1. Children under 2 years of age. Pregnancy and breast-feeding (see section 4.6).
Side effects
Summary of the safety profileThe adverse events most frequently associated with oral administration of N-acetylcysteine are gastrointestinal in nature. Hypersensitivity reactions including anaphylactic shock, anaphylactic / anaphylactoid reactions, bronchospasm, angioedema, rash and pruritus have been reported less frequently.Tabular list of adverse reactionsAdverse reactions listed by classification system and frequency are presented in the following table: very common (≥ 1/10), common (≥ 1/100 to
| Organ-systemic classification | Adverse reactions |
| Uncommon (≥1 / 1,000; | Rare (≥1 / 10,000; | Very rare ( | Not known |
| Disorders of the immune system | Hypersensitivity | | Anaphylactic shock, anaphylactic / anaphylactoid reaction | |
| Nervous system disorders | Headache | | | |
| Ear and labyrinth disorders | Tinnitus | | | |
| Cardiac pathologies | Tachycardia | | | |
| Vascular pathologies | | | Hemorrhage | |
| Respiratory, thoracic and mediastinal disorders | | Bronchospasm, dyspnoea | | Bronchial obstruction |
| Gastrointestinal disorders | Vomiting, diarrhea, stomatitis, abdominal pain, nausea | Dyspepsia | | |
| Skin and subcutaneous tissue disorders | Urticaria, rash, angioedema, pruritus | | | |
| General disorders and administration site conditions | Pyrexia | | | Edema of the face |
| Diagnostic tests | Reduced blood pressure | | | |
Description of some adverse reactionsIn very rare cases, severe skin reactions have occurred in temporal connection with the intake of N-acetylcysteine, such as Stevens-Johnson syndrome and Lyell syndrome. Although in most cases at least one other suspected drug more likely involved in the genesis of the aforementioned mucocutaneous syndromes has been identified, in case of mucocutaneous alterations it is advisable to consult your doctor and the intake of N-acetylcysteine must be stopped immediately. Some studies have confirmed a reduction in platelet aggregation when taking N-acetylcysteine. The clinical significance of these findings has not yet been defined.Reporting of suspected adverse reactionsReporting of suspected adverse reactions that occur after authorization of the medicine is important, as it allows continuous monitoring of the benefit / risk ratio of the medicine. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system at https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse
Special warnings
Patients with bronchial asthma should be closely monitored during therapy, if bronchospasm occurs, treatment with N-acetylcysteine should be discontinued immediately and appropriate treatment initiated. Mucolytics can induce bronchial obstruction in children under 2 years of age. In fact, the drainage capacity of the bronchial mucus is limited in this age group, due to the physiological characteristics of the respiratory tract. They should therefore not be used in children less than 2 years of age (see section 4.3). The use of the medicinal product in patients with peptic ulcer or with a history of peptic ulcer requires particular attention, especially in the case of concomitant use of other drugs with a known gastrically damaging effect. The possible presence of a sulphurous odor does not indicate alteration of the preparation but is typical of the active ingredient contained therein. The administration of N-acetylcysteine, especially at the beginning of the treatment, can thin the bronchial secretions and at the same time increase their volume. If the patient is unable to expectorate effectively, postural drainage and bronchoaspiration should be used to avoid secretion retention. N-acetylcysteine can affect histamine metabolism. Therefore, caution should be used when administering Mucolytic Fluimucil to patients with histamine intolerance, as hypersensitivity symptoms may occur. Important information about some of the ingredients Sodium benzoate 100 mg / 5 ml syrup (150 ml and 200 ml) contains 15 mg of sodium benzoate for the 10 ml dose and 7.5 mg for the 5 ml dose.ParahydroxybenzoatesThe syrups contain parahydroxybenzoates which can cause delayed allergic reactions.SorbitolThe buccal tablets, 600 mg / 15 ml syrup, 600 mg granules for oral solution and sugar-free granules for oral solution (100 mg and 200 mg) contain sorbitol. The content of sorbitol in oral medicinal products may modify the bioavailability of other co-administered oral medicinal products. Patients with hereditary fructose intolerance should not be given these medicines.AspartameThe buccal tablets, effervescent tablets, 600 mg granules for oral solution and 100 and 200 mg sugar-free granules for oral solution contain aspartame, a source of phenylalanine which may be harmful in patients with phenylketonuria.GlucoseThe effervescent tablets of 600 mg, the granules for oral solution of 600 mg and the granules for oral solution of 200 mg contain glucose, patients with rare problems of glucose-galactose malabsorption should not take this medicine.Sunset Yellow (E110)The 100 mg and 200 mg granules for oral solution contain sunset yellow (E110) which may cause allergic reactions.SucroseThe 100 and 200 mg granules for oral solution contain sucrose. Patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption or sucrase isomaltase insufficiency should not take this medicine.SodiumThe buccal tablets contain 26.9 mg of sodium per tablet equivalent to 1.3% of the WHO recommended maximum daily intake of 2 g of sodium for an adult. The 200 mg and 600 mg effervescent tablets contain 156.9 mg of sodium per dose, equivalent to 7.8% of the WHO recommended maximum daily intake of 2 g of sodium for an adult. The 100mg / 5ml (150ml) syrup contains 36.7mg sodium per 10ml serving, equivalent to 1.83% of the WHO recommended maximum daily intake of 2g sodium for an adult . The 100 mg / 5 ml (150 ml) syrup contains 18.4 mg of sodium per 5 ml dose, equivalent to 0.9% of the WHO recommended maximum daily intake of 2 g of sodium for an adult . The 100mg / 5ml (200ml) syrup contains 38.2mg sodium per 10ml serving, equivalent to 1.9% of the WHO recommended maximum daily intake of 2g sodium for an adult. The 100 mg / 5 ml (200 ml) syrup contains 19.1 mg of sodium for the 5 ml dose, equivalent to 0.9% of the WHO recommended maximum daily intake which corresponds to 2 g of sodium for one adult. The 600mg / 15ml syrup contains 98.31mg sodium per 15ml dose equivalent to 4.9% of the WHO recommended maximum daily intake of 2g sodium for an adult.LactoseThe 600 mg granules for oral solution and the 200 mg granules for oral solution contain lactose. Patients with rare hereditary problems of galactose intolerance, total lactase deficiency, or glucose-galactose malabsorption should not take this medicine.Propylene glycolThe 100 mg / 5 ml syrup (150 ml and 200 ml) contains 23.4 mg of propylene glycol for the 10 ml dose and 11.7 mg for the 5 ml dose. The 600 mg / 15 mL syrup contains 168 mg propylene glycol per dose (15 mL) equivalent to 11.2 mg / mL.EthanolThe 100mg / 5ml syrup (150ml and 200ml) contains 3.85mg of alcohol (ethanol) in each 100ml. The dose quantity of this medicine is equivalent to less than 1 ml of beer or 1 ml of wine. The small amount of alcohol in this medicine will not produce relevant effects.
Pregnancy and breastfeeding
Even if the teratological studies conducted with Fluimucil Mucolytic on animals have not shown any teratogenic effect, however, as for other drugs, its administration during pregnancy and during the period of breastfeeding with breast milk, should be carried out only in case of actual need. .
Expiration and retention
Sachets of 100 and 200 granules for oral solution, 600 mg granules for oral solution, 200 mg granules for oral solution without sugar and 200 mg buccal tablets: store at a temperature not exceeding 30 ° C.
Interactions with other drugs
Drug-drug interaction. Antitussive drugs and mucolytic agents, such as N-acetylcysteine, should not be taken at the same time as the reduced cough reflex could lead to an accumulation of bronchial secretions. Activated carbon can reduce the effect of N-acetylcysteine. It is advisable not to mix other drugs with Fluimucil Mucolytic solution. The information available on the antibiotic-N-acetylcysteine interaction refers to in vitro tests, in which the two substances were mixed, which showed a decreased activity of the antibiotic. However, as a precaution, it is recommended to take antibiotics by mouth at least two hours after the administration of N-acetylcysteine with the exception of loracarbef. It has been shown that the simultaneous intake of nitroglycerin and N-acetylcysteine causes significant hypotension and causes dilation of the temporal artery with possible onset of headache. If the simultaneous administration of nitroglycerin and N-acetylcysteine is necessary, patients should be monitored for the onset of hypotension, which can also be severe, and warned about the possible onset of headache.Pediatric populationInteraction studies have only been performed in adults.Drug-laboratory test interactionsN-acetylcysteine can cause interference with the colorimetric assay method for the determination of salicylates. N-acetylcysteine can interfere with the test for the determination of ketones in urine.
Overdose
No cases of overdose have been reported with respect to oral administration of N-acetylcysteine. Healthy volunteers who took a daily dose of N-acetylcysteine of 11.6 g for three months did not show any serious adverse reactions. Doses up to 500 mg NAC / kg body weight, administered orally, were tolerated without any symptoms of intoxication.SymptomsOverdose can cause gastrointestinal symptoms such as nausea, vomiting and diarrhea.TreatmentThere are no specific antidotic treatments; overdose therapy is based on symptomatic treatment.
Active principles
FLUIMUCIL MUCOLITICO 600 mg effervescent tabletsEach tablet contains:Active principle: N-acetylcysteine 600 mg. Excipients with known effects: aspartame, glucose, sodium.FLUIMUCIL MUCOLITICO 600 mg granules for oral solutionEach sachet contains:Active principle: N-acetylcysteine 600 mg. Excipients with known effects: aspartame, glucose, lactose, sorbitol.FLUIMUCIL MUCOLITICO 600 mg / 15 ml syrup15 ml of syrup contain:Active principle: N-acetylcysteine 600 mg. Excipients with known effects: propylene glycol, methyl para-hydroxybenzoate, propyl para-hydroxybenzoate, sodium, sorbitol.FLUIMUCIL MUCOLITICO 200 mg, effervescent tabletsOne tablet contains:Active principle: N-acetylcysteine 200 mg. Excipients with known effects: sodium, aspartame.FLUIMUCIL MUCOLITICO 200 mg, buccal tablets:One tablet contains:Active principle: N-acetylcysteine 200 mg. Excipients with known effects: sorbitol, sodium, aspartame.FLUIMUCIL MUCOLITICO 200 mg, granules for oral solutionOne sachet contains:Active principle: N-acetylcysteine 200 mg. Excipients with known effects: sucrose, glucose, sunset yellow (E110), lactose.FLUIMUCIL MUCOLITICO 200 mg, granules for oral solution without sugarOne sachet contains:Active principle: N-acetylcysteine 200 mg. Excipients with known effects: sorbitol, aspartame.FLUIMUCIL MUCOLITICO 100 mg, granules for oral solutionOne sachet contains:Active principle: N-acetylcysteine 100 mg. Excipients with known effects: sucrose, sunset yellow (E110).FLUIMUCIL MUCOLITICO 100 mg, granules for oral solution without sugarOne sachet contains:Active principle: N-acetylcysteine 100 mg. Excipients with known effects: sorbitol, aspartame.FLUIMUCIL MUCOLITICO 100 mg / 5 ml, syrupA 150ml bottle contains:Active principle: N-acetylcysteine 3,000 g (corresponding to 100 mg / 5 ml of syrup). Excipients with known effects: ethanol, methyl parahydroxybenzoate, propylene glycol, sodium benzoate, sodium. A 200ml bottle contains:Active principle: N-acetylcysteine 4,000 g (corresponding to 100 mg / 5 ml of syrup). Excipients with known effects: ethanol, methyl parahydroxybenzoate, propylene glycol, sodium benzoate, sodium. For the full list of excipients, see section 6.1.
Excipients
FLUIMUCIL MUCOLITICO 600 mg granules for oral solutionAspartame, Orange flavor (containing glucose and lactose), Sorbitol.FLUIMUCIL MUCOLITICO 600 mg effervescent tabletsAnhydrous citric acid, Lemon flavor (containing glucose), Aspartame, Sodium bicarbonate.FLUIMUCIL MUCOLITICO 600 mg / 15 ml syrup 200 ml bottleMethyl parahydroxybenzoate, Propyl parahydroxybenzoate, Sodium edetate, Carmellose, Saccharin sodium, Grenadine flavor (containing propylene glycol), Strawberry flavor (containing propylene glycol), Sorbitol, Sodium hydroxide, Purified water.FLUIMUCIL MUCOLITICO 200 mg buccal tabletsAnhydrous citric acid, sorbitol, mannitol, polyethylene glycol 6000, povidone, sodium bicarbonate, lemon flavor, mandarin flavor, aspartame, magnesium stearate, microcrystalline cellulose.FLUIMUCIL MUCOLITICO 200 mg granules for oral solution without sugarSorbitol, aspartame, orange flavor.FLUIMUCIL MUCOLITICO 200 mg granules for oral solutionOrange juice granules; Orange flavor (containing glucose and lactose); saccharin; sunset yellow (E 110); Sucrose.FLUIMUCIL MUCOLITICO 200 mg effervescent tablets Anhydrous citric acid, sodium bicarbonate, lemon flavor, aspartame. FLUIMUCIL MUCOLITICO 100 mg granules for oral solutionOrange juice granules; Orange flavor; Saccharin; E 110; Sucrose.FLUIMUCIL MUCOLITICO 100 mg granules for oral solution without sugarSorbitol; Aspartame; Orange flavor.FLUIMUCIL MUCOLITICO 100 mg / 5 ml syrup 150 ml bottleMethyl parahydroxybenzoate, sodium benzoate, sodium edetate, sodium carboxymethylcellulose, raspberry flavor (containing propylene glycol and ethanol), sodium saccharinate, sodium hydroxide, purified water.FLUIMUCIL MUCOLITICO 100 mg / 5 ml syrup 200 ml bottleMethyl parahydroxybenzoate, sodium benzoate, sodium edetate, sodium carboxymethylcellulose, sodium cyclamate, sucralose, raspberry flavor (containing propylene glycol and ethanol), sodium saccharinate, sodium hydroxide, purified water.

 Convenient and safe payments
Convenient and safe payments
 Shipping in 24h all over the world
Shipping in 24h all over the world
 Fast returns
Fast returns
 Pharmaceutical experience
Pharmaceutical experience